KR320P (Peroxide Curable Terpolymer)
FKM (Fluorocarbon Rubber or Fluoroelastomer) Raw material
Technical Information
FLUONOX® KR 320P is a low viscosity peroxide curable 67% fluorine terpolymer. It can be cross-linked using organic peroxides in combination with coagent. FLUONOX® KR320P can be used for injection and transfer moulding of shaft seals, O-rings and gaskets and extrusion of hosesand profiles.
Product features
* Good chemical resistance
* Good mould release
* Good extrusion behavior
* Good low temperature performance
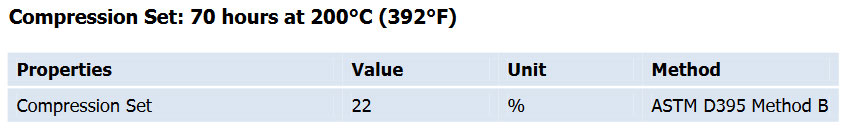
* Good compression set
Packaging
Fluonox®KR320Pis available in 25kg box.

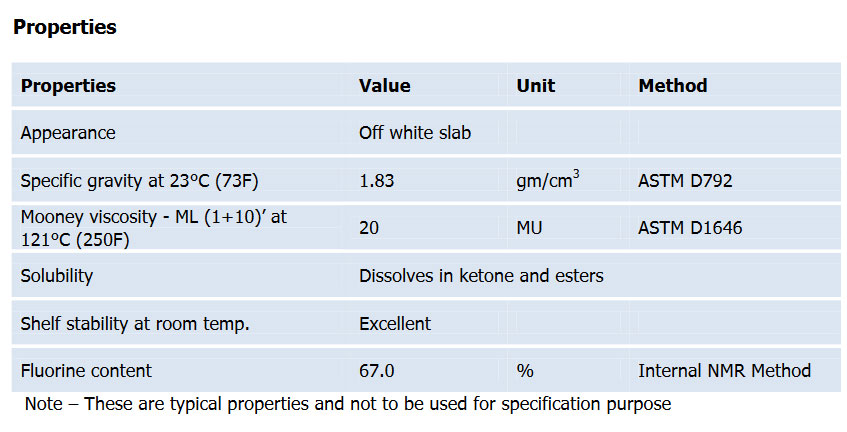
Physical properties:
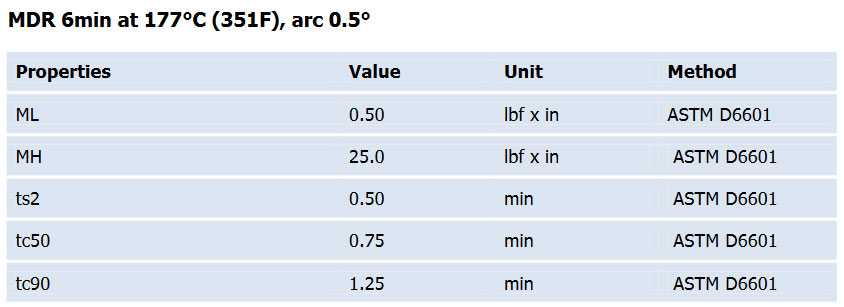
Press cure 10 min at 170°C(338F); Post cure 4 hours at 230°C(446F)
Safety and Handling
Handling and processing of fluoroelastomer must be done in ventilated areas to prevent personnel exposure to the fumes liberated during
curing or use of cured rubber at high temperature. During the process, some fumes may generate at high temperature which are harmful
for human beings. Fumes should not be inhaled; eye and skin contact must be avoided. In case of skin contact flush skin with cold water
immediately. In case of eye contact, flush with water immediately and seek medical help. Smoking tobacco or cigarettes should not be
allowed in working area. Mixing agents that contain metallic particulate such as powdered aluminium can rapidly decompose at high
temperature; therefore do not use metallic particulate as mixing agent. Fluoroelastomer should be stored away from heat. It should be kept
in clean and dry area where it can be protected until it is used. Please read the Material Safety Data Sheet before handling the product.
Disclaimer
FLUONOX® is the brand name of Gujarat Fluorochemicals Limited (GFL) used for its brand of fluoroelastomer. FLUONOX® can be used in
applications duly approved by GFL. Customers who plan to use the word FLUONOX® as the trade mark on or relation to their own
fluoroelastomer parts and other products in any style or combination or in any manner whatsoever must contact GFL for prior permission
for such use. No consumer/user of GFL fluoropolymer resin is permitted to claim that their products contain FLUONOX®without prior
permission from GFL.
The information provided in the bulletin is furnished at no cost to the recipient and is based on information and technical data that Gujarat
Fluorochemicals Limited believes is correct and sound. Those who choose to use the information must be technically qualified, and do so
entirely at their own cost and risk. The users must determine and insure that their specific conditions of processing present no health or
safety hazards. GFL does not warranty, either expressly or impliedly in respect of use of this information for application of its FLUONOX®
branded Fluoroelastomer and shall bear no liability as a result of any loss or damage caused directly or indirectly due to use of any
information provided in this bulletin. Nothing contained herein can be taken or construed as a grant of license by GFL to operate under or a
recommendation to infringe any patents.
Note warning
Do not use any of FLUONOX® Fluoroelastomer in medical devices that are designed for permanent implantation in the human body. For
other medical uses, prior permission of GFL may be sought.
